Primary cutaneous aggressive epidermotropic CD8+ T-cell lymphoma: Transformation from indolent to aggressive phase in association with CCR7-positive conversion
Published Web Location
https://doi.org/10.5070/D37410r9g1Main Content
Primary cutaneous aggressive epidermotropic CD8+ T-cell lymphoma: Transformation from indolent to aggressive phase in association
with CCR7-positive conversion
Akira Kasuya MD, Satoshi Hirakawa MD PhD, Yoshiki Tokura
Dermatology Online Journal 18 (10): 5
Hamamatsu University School of Medicine Hamamatsu, Shizuoka, JapanAbstract
We report a case of primary cutaneous aggressive epidermotropic CD8+ T-cell lymphoma that transformed from the indolent phase to the aggressive phase. In the aggressive phase, the patient developed numerous subcutaneous lesions on the trunk, limbs, and face. The involvement of muscle, right testis, and pharynx were also found. We immunohistochemically analyzed the expression of chemokine receptors, before and after the transformation of aggressive epidermotropic CD8+ T-cell lymphoma. The transformation was accompanied by positive conversion of CCR7. CCR7 is crucial in lymphatic cell migration and chemotaxis to lymph nodes. Therefore, its expression is likely associated with the disseminated behavior of T-cell lymphoma.
Introduction
Cutaneous T-cell lymphoma occasionally exhibits transition from the indolent to the aggressive phase. However, there are only a few studies on the conversion of the chemokine receptor profile of tumor cells [1]. We immunohistochemically analyzed the expression of chemokine receptors, before and after the transformation of aggressive epidermotropic CD8+ T-cell lymphoma.
Case report
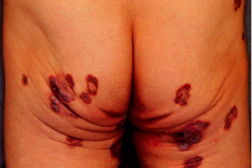
An 85-year-old man had been suffering from indolent primary cutaneous CD8+ T-cell lymphoma for 8 years. The initial lesions were multiple, discrete, violaceous plaques and tumors on the trunk with or without ulcerations (Figure 1).
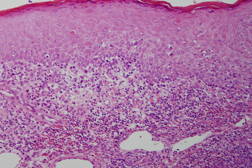
There were no parapsoriatic lesions suggestive of mycosis fungoides. Neither swollen superficial lymph nodes nor visceral lesions were detected by computed tomography (CT). The patient’s serum was negative for anti-HTLV-1 antibodies. Skin biopsy showed massive infiltration of atypical, small to medium-sized lymphocytes in the dermis with epidermotropism (Figure 2).
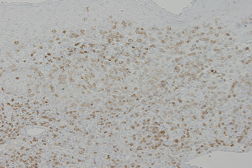
The atypical cells were immunohistochemically positive for CD2, CD3, CD8 (Figure 3), TIA, granzyme B and CXCR3, and negative for CD4, CD7, CD20, CD56, CD79a, CXCR4, CCR4 and CCR7. PCR analysis confirmed clonal gene rearrangement of the T-cell receptor Cβ1.
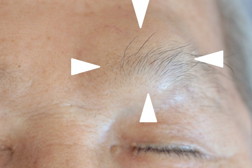
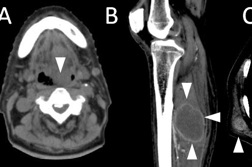
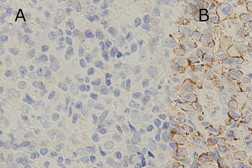
The initial clinical course was indolent, as one or two plaques newly appeared at an interval of a few months. These lesions responded well to radiation therapy or narrow-band UVB. Eight years after the onset, however, he developed numerous subcutaneous lesions on the trunk, limbs, and face (Figure 4). The involvement of muscle, right testis and pharynx was found on CT examination (Figure 5). Laryngeal endoscopy showed a nodule on his pharynx, 25 mm in diameter. Pharyngeal biopsy revealed an infiltrate of medium to large-sized lymphocytes with obvious nuclear atypia. The tumor cells were immunohistochemically same as those of the previous skin lesion, however, CCR7 was converted from negative to positive (Figure 6).
CD30, ALK, EBER, and T-cell receptor γδ were negative. Based on the aggressive clinical behavior, multiorgan involvement, and cytotoxic immunophenotype, we diagnosed the lesions as aggressive epidermotropic CD8+ T-cell lymphoma. Although CD2 positivity is inconsistent with the aggressive epidermotropic CD8+ T-cell lymphoma, its absence is not essential for the diagnosis of this lymphoma. Berti et al reported that 2 out of 8 cases of aggressive epidermotropic T-cell lymphoma were negative for CD2 [2]. After radiation therapy, 30 Gry/10 fraction, the lesion was markedly scaled down in a week. One month later, however, the patient died of cardio-respiratory failure.
Transformation to the aggressive form has been occasionally documented in CD8+ cutaneous lymphomas [3]. Ito et al reported a case of CD8+ cutaneous lymphoma showing a 2-year indolent course followed by transformation into the aggressive phase [3]. Because the chemokine receptors, CXCR3, CXCR4, CCR4 and CCR7, have been proposed to be involved in the behavior of cutaneous T-cell lymphoma [4], we performed immunostaining for these.
CCR4 is a skin homing chemokine. In cutaneous T-cell lymphoma, e.g. mycosis fungoides and Sézary syndrome, malignant lymphocytes are reported to be positive for CCR4. The expression of CCR4 might be involved in recruitment of malignant lymphocytes to the skin. CCR7 is reported to be positive in Sézary syndrome and negative in mycosis fungoides [5, 6]. CCR7 is crucial in lymphocyte migration and chemotaxis to lymph nodes [7]. Therefore, its expression might be associated with the aggressive behavior of Sézary syndrome.
We found that CCR7converted to positive in the pharynx lesion of the aggressive phase, whereas CCR4 remained negative. The phenotype of CCR7+CCR4- might explain the disseminated behavior and decreased epidermotropism in our case.
Conclusion
We found the occurrence of CCR7-positive conversion in the process of transformation from the indolent to the aggressive phase in primary cutaneous aggressive epidermotropic CD8+ T-cell lymphoma. CCR7-positive conversion might partly contribute to the aggressive behavior of tumor in our case.
References
1. Tokura Y, Sugita K, Yagi H, Shimauchi T, Kabashima K, Takigawa M. Primary cutaneous anaplastic large cell lymphoma with fatal leukemic outcome in association with CLA and CCR4-negative conversion. J Am Acad Dermatol 2007 Nov;57(5 Suppl):S92-6. [PubMed]2. Berti E, Tomasini D, Vermeer MH, Meijer CJ, Alessi E, Willemze R. Primary cutaneous CD8-positive epidermotropic cytotoxic T cell lymphomas. A distinct clinicopathological entity with an aggressive clinical behavior. Am J Pathol 1999 Aug;155(2):483-92. [PubMed]
3. Ito Y, Goto M, Hatano Y, Kawamoto M, Ohishi M, Takayasu S, Katagiri K, Fujiwara S. Epidermotropic CD8+ cytotoxic T-cell lymphoma exhibiting a transition from the indolent to the aggressive phase, accompanied by emergence of CD7+ cells and formation of neutrophilic pustules. Clin Exp Dermatol. 2012 Mar;37(2):128-31. [PubMed]
4. Wu XS, Lonsdorf AS, Hwang ST. Cutaneous T cell lymphoma: Roles for chemokines and chemokine receptors. J Invest Dermatol 2009 May;129(5):1115-9. [PubMed]
5. Campbell JJ, Clark RA, Watanabe R, Kupper TS. Sézary syndrome and mycosis fungoides arise from distinct T-cell subsets: a biologic rationale for their distinct clinical behaviors. Blood. 2010 Aug 5;116(5):767-71. [PubMed]
6. Sokolowska-Wojdylo M, Wenzel J, Gaffal E, Lenz J, Speuser P, Erdmann S, Abuzahra F, Bowman E, Roszkiewicz J, Bieber T, Tüting T. Circulating clonal CLA(+) and CD4(+) T cells in Sézary syndrome express the skin-homing chemokine receptors CCR4 and CCR10 as well as the lymph node-homing chemokine receptor CCR7. Br J Dermatol. 2005 Feb;152(2):258-64. [PubMed]
7. Yang J, Wang S, Zhao G, Sun B. Effect of chemokine receptors CCR7 on disseminate behavior of human T cell lymphoma: clinical and experimental study. J Exp Clin Cancer Res 2011 May 7;30:51. [PubMed]
© 2012 Dermatology Online Journal